CSD & Migraine with Aura
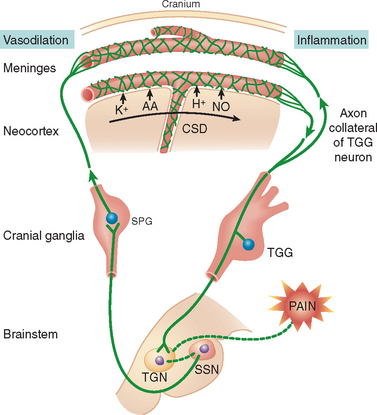
In about a third of migraine patients, focal neurological disturbances known as auras are experienced as visual, motor, or sensory symptoms. Many studies have established aura in humans is analogous to CSD of Leão in other animals (Charles & Baca, 2013). CSD, cerebral spreading depression, is a wave of activity that moves across the surface of the brain slowly, as described in several diverse animal models ( Leão, 1944a, 1944b, 1947; Leão & Morison, 1945). For instance, a wave of depolarization was observed as a massive shift in direct current (DC) which was then followed by a spreading suppression or “depression” of spontaneous EEG activity. The wave of depolarization is seen in glial cells and neurons with a great rise in extracellular K+, decrease in extracellular Na+, among other changes in ion concentrations (Hansen & Zeuthen, 1981; Dietz et al., 2008; Mutch & Hansen, 1984). These ion fluxes can cause cellular swelling that may change the extracellular environment. Almost all areas of the brain can exhibit CSD (Leão, 1947). Visual aura in humans alters the visual field that begins at the center of the field and moves to the outside at 3 mm/min (Goadsby, 2013). This phenomenon has been mapped onto areas believed to correspond to the occipital cortex which resembles the spatial and temporal aspects of CSD in rabbits (Charles & Baca, 2013). In MA patients, spatial patterns of DC switching was observed when using magnetoencephalography that proposes the involvement of CSD (Bowyer et al., 2001). Sustained aura can be alleviated by ketamine, a validated agent for blocking CSD in animals (Goadsby, 2013). However, the standard electrophysiological characteristics of CSD have not detected in subjects who experience migraine. Further, there were no significant changes in surface EEG recordings with scalp electrodes while aura was occurring (Lauritzen et al., 1981). In migraine patients, functional imaging studies have shown changes in blood flow and cortical activity, with congruous temporal and spatial features to CSD. CT images of MA-induced in patients exhibited a wave of oligaemia, then hyperaemia, in large regions of the center (Olesen et al., 1990). A case study of an MO patient using positron emission tomography, PET, also displayed oligaemia when a migraine headache present (Woods et al., 1994). CSD is indicated in migraine since the changes in blood flow and cortical function in patients with MO and MA show a pattern of propagation throughout the brain. Another debate is whether pain is experienced with CSD. In animal models, pain pathways may be activated or sensitized when messengers, such as nitric oxide (Colonna et al., 1997; Obrenovitch et al., 2002), ATP (Schock et al., 2007), CGRP (Colonna et al., 1994; Reuter et al., 1998; Tozzi et al., 2012; Wahl et al., 1994), and high-mobility group protein B1 (Karatas et al., 2013), are released while CSD is occurring. Electrophysiological neuronal recordings in the trigeminal ganglion and brainstem, along with immunohistological c-Fos labelling in the brain stem, demonstrate trigeminal nociceptive neuron activation during CSD (Zhang et al., 2010, 2011). However, Nmethyl- D-aspartate given to awake rats causes a CSD occurrence and freezing behavior, but not ultrasonic vocalization associated with extreme pain (Akcali et al., 2010). A different study observed facial expressions indicating pain when KCl was employed to evoke CSD (Karatas et al., 2013).
CSD & Migraine with Aura

